Drug formerly under development for treatment of menopause symptoms
Pharmaceutical compound
Esmirtazapine Routes of Oral ATC code Legal status Metabolism Liver (CYP2D6 )[ 2] Elimination half-life 10 hours[ 1]
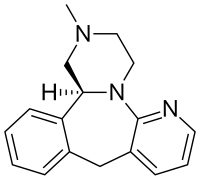
(S )-1,2,3,4,10,14b-hexahydro-2-methylpyrazino(2,1-a)pyrido(2,3-c)(2)benzazepine
CAS Number PubChem CID DrugBank ChemSpider UNII KEGG CompTox Dashboard (EPA ) ECHA InfoCard 100.056.994 Formula C 17 H 19 N 3 Molar mass −1 3D model (JSmol ) Melting point 114 to 116 °C (237 to 241 °F) Solubility in water Soluble in methanol and chloroform
CN1CCN2c3c(cccn3)Cc4ccccc4[C@H]2C1
InChI=1S/C17H19N3/c1-19-9-10-20-16(12-19)15-7-3-2-5-13(15)11-14-6-4-8-18-17(14)20/h2-8,16H,9-12H2,1H3/t16-/m1/s1
Y Key:RONZAEMNMFQXRA-MRXNPFEDSA-N
Y N Y (what is this?) (verify)
Esmirtazapine (ORG-50,081 ) is a tetracyclic antidepressant drug that was under development by Organon for the treatment of insomnia and vasomotor symptoms (e.g., hot flashes ) associated with menopause .[ 3] [ 4] [ 5] [ 6] S )-(+)-enantiomer of mirtazapine and possesses similar overall pharmacology , including inverse agonist actions at H1 and 5-HT2 receptors and antagonist actions at α2 -adrenergic receptors .[ 3] [ 7]
Notably, esmirtazapine has a shorter half life of around 10 hours, compared to R-mirtazapine and racemic mixture, which has a half-life of 18–40 hours.[ 1] 1 -inhibitor and a 5-HT2A antagonist.[ 8] [ 1] [ 9]
In March 2010, Merck terminated its internal clinical development program for esmirtazapine for hot flashes and insomnia, "for strategic reasons".[ 10]
See also
References
^ a b c Ruwe F, IJzerman-Boon P, Roth T, Zammit G, Ivgy-May N (October 2016). "A Phase 2 Randomized Dose-Finding Study With Esmirtazapine in Patients With Primary Insomnia". Journal of Clinical Psychopharmacology . 36 (5): 457– 464. doi :10.1097/JCP.0000000000000546 . PMID 27482970 . S2CID 25639396 . ^ Lillin-de Vries O, Kerbusch T, de Kam PJ, Ivgy-May N, Bursi R. "A population analysis on the effects of the CYP2D6 deficiency on pharmacokinetics and exposure of esmirtazapine in healthy volunteers" (PDF) . Oss, The Netherlands: Schering-Plough Corporation. ^ a b "Future Treatments for Depression, Anxiety, Sleep Disorders, Psychosis, and ADHD" . Neurotransmitter.net .^ Clinical trial number NCT00561574 ClinicalTrials.gov
^ Teegarden BR, Al Shamma H, Xiong Y (2008). "5-HT(2A) inverse-agonists for the treatment of insomnia". Current Topics in Medicinal Chemistry . 8 (11): 969– 976. doi :10.2174/156802608784936700 . PMID 18673166 . ^ Lewis V (November 2009). "Undertreatment of menopausal symptoms and novel options for comprehensive management". Current Medical Research and Opinion . 25 (11): 2689– 2698. doi :10.1185/03007990903240519 . PMID 19775194 . S2CID 206964530 . ^ Stahl SM (2008). "Antidepresents" . Depression and bipolar disorder: Stahl's essential psychopharmacology (3rd ed.). Cambridge, UK: Cambridge University Press. ISBN 978-0-521-88663-5 ^ Ivgy-May N, Ruwe F, Krystal A, Roth T (July 2015). "Esmirtazapine in non-elderly adult patients with primary insomnia: efficacy and safety from a randomized, 6-week sleep laboratory trial". Sleep Medicine . 16 (7): 838– 844. doi :10.1016/j.sleep.2015.04.001 . PMID 26047892 . ^ Ivgy-May N, Hajak G, van Osta G, Braat S, Chang Q, Roth T (September 2020). "Efficacy and safety of esmirtazapine in adult outpatients with chronic primary insomnia: a randomized, double-blind placebo-controlled study and open-label extension" . Journal of Clinical Sleep Medicine . 16 (9): 1455– 1467. doi :10.5664/jcsm.8526 . PMC 7970588 PMID 32351205 . ^ "Form 10-K" (PDF) . Merck & Co., Inc. Archived from the original (PDF) on 2011-08-05. Retrieved 2011-05-03 .
External links
SSRIs Tooltip Selective serotonin reuptake inhibitors SNRIs Tooltip Serotonin–norepinephrine reuptake inhibitors NRIs Tooltip Norepinephrine reuptake inhibitors NDRIs Tooltip Norepinephrine–dopamine reuptake inhibitors NaSSAs Tooltip Noradrenergic and specific serotonergic antidepressants SARIs Tooltip Serotonin antagonist and reuptake inhibitors SMS Tooltip Serotonin modulator and stimulators Others
TCAs Tooltip Tricyclic antidepressants TeCAs Tooltip Tetracyclic antidepressants Others
Non-selective MAOA Tooltip Monoamine oxidase A -selectiveMAOB Tooltip Monoamine oxidase B -selective
α1
Agonists Antagonists
Abanoquil Ajmalicine Alfuzosin Anisodamine Anisodine Atiprosin Atypical antipsychotics (e.g., brexpiprazole , clozapine , olanzapine , quetiapine , risperidone )Benoxathian Beta blockers (e.g., adimolol , amosulalol , arotinolol , carvedilol , eugenodilol , labetalol )Buflomedil Bunazosin Corynanthine Dapiprazole Domesticine Doxazosin Ergolines (e.g., acetergamine , ergotamine , dihydroergotamine , lisuride , nicergoline , terguride )Etoperidone Fenspiride Hydroxyzine Indoramin Ketanserin L-765,314 mCPP Mepiprazole Metazosin Monatepil Moxisylyte Naftopidil Nantenine Neldazosin Niaprazine Niguldipine Pardoprunox Pelanserin Perlapine Phendioxan Phenoxybenzamine Phentolamine Phenylpiperazine antidepressants (e.g., hydroxynefazodone , nefazodone , trazodone , triazoledione )Piperoxan Prazosin Quinazosin Quinidine Silodosin Spegatrine Spiperone Talipexole Tamsulosin Terazosin Tiodazosin Tolazoline Tetracyclic antidepressants (e.g., amoxapine , maprotiline , mianserin )Tricyclic antidepressants (e.g., amitriptyline , clomipramine , doxepin , imipramine , trimipramine )Trimazosin Typical antipsychotics (e.g., chlorpromazine , fluphenazine , loxapine , thioridazine )Urapidil WB-4101 Zolertine
α2
Agonists Antagonists
1-PP Adimolol Amesergide Aptazapine Atipamezole Atypical antipsychotics (e.g., asenapine , brexpiprazole , clozapine , lurasidone , olanzapine , paliperidone , quetiapine , risperidone , zotepine )Azapirones (e.g., buspirone , gepirone , ipsapirone , tandospirone )BRL-44408 Buflomedil Cirazoline Efaroxan Esmirtazapine Fenmetozole Fluparoxan Idazoxan Ketanserin Lisuride mCPP Mianserin Mirtazapine NAN-190 Pardoprunox Phentolamine Phenoxybenzamine Piperoxan Piribedil Rauwolscine Rotigotine Setiptiline Spegatrine Spiroxatrine Sunepitron Terguride Tolazoline Typical antipsychotics (e.g., chlorpromazine , fluphenazine , loxapine , thioridazine )Yohimbine
β
H1
Agonists Antagonists
Others: Atypical antipsychotics (e.g., aripiprazole , asenapine , brexpiprazole , brilaroxazine , clozapine , iloperidone , olanzapine , paliperidone , quetiapine , risperidone , ziprasidone , zotepine )Phenylpiperazine antidepressants (e.g., hydroxynefazodone , nefazodone , trazodone , triazoledione )Tetracyclic antidepressants (e.g., amoxapine , loxapine , maprotiline , mianserin , mirtazapine , oxaprotiline )Tricyclic antidepressants (e.g., amitriptyline , butriptyline , clomipramine , desipramine , dosulepin (dothiepin) , doxepin , imipramine , iprindole , lofepramine , nortriptyline , protriptyline , trimipramine )Typical antipsychotics (e.g., chlorpromazine , flupenthixol , fluphenazine , loxapine , perphenazine , prochlorperazine , thioridazine , thiothixene )
H2
H3
H4
5-HT1
5-HT1A
Agonists: 8-OH-DPAT Adatanserin Amphetamine Antidepressants (e.g., etoperidone , hydroxynefazodone , nefazodone , trazodone , triazoledione , vilazodone , vortioxetine )Atypical antipsychotics (e.g., aripiprazole , asenapine , brexpiprazole , cariprazine , clozapine , lurasidone , quetiapine , ziprasidone )Azapirones (e.g., buspirone , eptapirone , gepirone , perospirone , tandospirone )Bay R 1531 Befiradol BMY-14802 Cannabidiol Dimemebfe Dopamine Ebalzotan Eltoprazine Enciprazine Ergolines (e.g., bromocriptine , cabergoline , dihydroergotamine , ergotamine , lisuride , LSD , methylergometrine (methylergonovine) , methysergide , pergolide )F-11,461 F-12826 F-13714 F-14679 F-15063 F-15,599 Flesinoxan Flibanserin Flumexadol Hypidone Lesopitron LY-293284 LY-301317 mCPP MKC-242 Naluzotan NBUMP Osemozotan Oxaflozane Pardoprunox Piclozotan Rauwolscine Repinotan Roxindole RU-24,969 S-14,506 S-14671 S-15535 Sarizotan Serotonin (5-HT) SSR-181507 Sunepitron Tryptamines (e.g., 5-CT , 5-MeO-DMT , 5-MT , bufotenin , DMT , indorenate , N-Me-5-HT , psilocin , psilocybin )TGBA01AD U-92,016-A Urapidil Vilazodone Xaliproden Yohimbine
Antagonists: Atypical antipsychotics (e.g., iloperidone , risperidone , sertindole )AV965 Beta blockers (e.g., alprenolol , carteolol , cyanopindolol , iodocyanopindolol , isamoltane , oxprenolol , penbutolol , pindobind , pindolol , propranolol , tertatolol )BMY-7,378 CSP-2503 Dotarizine Ergolines (e.g., metergoline )FCE-24379 Flopropione GR-46611 Isamoltane Lecozotan Mefway Metitepine (methiothepin) MIN-117 (WF-516) MPPF NAN-190 Robalzotan S-15535 SB-649,915 SDZ 216-525 Spiperone Spiramide Spiroxatrine UH-301 WAY-100135 WAY-100635 Xylamidine
5-HT1B
Agonists: Anpirtoline CGS-12066A CP-93129 CP-94253 CP-122,288 CP-135807 Eltoprazine Ergolines (e.g., bromocriptine , dihydroergotamine , ergotamine , methylergometrine (methylergonovine) , methysergide , pergolide )mCPP RU-24,969 Serotonin (5-HT) Triptans (e.g., avitriptan , donitriptan , eletriptan , sumatriptan , zolmitriptan )TFMPP Tryptamines (e.g., 5-BT , 5-CT , 5-MT , DMT )Vortioxetine
5-HT1D
Agonists: CP-122,288 CP-135807 CP-286601 Ergolines (e.g., bromocriptine , cabergoline , dihydroergotamine , ergotamine , LSD , methysergide )GR-46611 L-694247 L-772405 mCPP PNU-109291 PNU-142633 Serotonin (5-HT) TGBA01AD Triptans (e.g., almotriptan , avitriptan , donitriptan , eletriptan , frovatriptan , naratriptan , rizatriptan , sumatriptan , zolmitriptan )Tryptamines (e.g., 5-BT , 5-CT , 5-Et-DMT , 5-MT , 5-(nonyloxy)tryptamine , DMT )
5-HT1E
5-HT1F
5-HT2
5-HT2A
Agonists: 25H/NB series (e.g., 25I-NBF , 25I-NBMD , 25I-NBOH , 25I-NBOMe , 25B-NBOMe , 25C-NBOMe , 25TFM-NBOMe , 2CBCB-NBOMe , 25CN-NBOH , 2CBFly-NBOMe )2Cs (e.g., 2C-B , 2C-E , 2C-I , 2C-T-2 , 2C-T-7 , 2C-T-21 )2C-B-FLY 2CB-Ind 5-Methoxytryptamines (5-MeO-DET , 5-MeO-DiPT , 5-MeO-DMT , 5-MeO-DPT , 5-MT )α-Alkyltryptamines (e.g., 5-Cl-αMT , 5-Fl-αMT , 5-MeO-αET , 5-MeO-αMT , α-Me-5-HT , αET , αMT )AL-34662 AL-37350A Bromo-DragonFLY Dimemebfe DMBMPP DOx (e.g., DOB , DOC , DOI , DOM )Efavirenz Ergolines (e.g., 1P-LSD , ALD-52 , bromocriptine , cabergoline , ergine (LSA) , ergometrine (ergonovine) , ergotamine , lisuride , LA-SS-Az , LSB , LSD , LSD-Pip , LSH , LSP , methylergometrine (methylergonovine) , pergolide )Flumexadol IHCH-7113 Jimscaline Lorcaserin MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )O-4310 Oxaflozane PHA-57378 PNU-22394 PNU-181731 RH-34 SCHEMBL5334361 Phenethylamines (e.g., lophophine , mescaline )Piperazines (e.g., BZP , quipazine , TFMPP )Serotonin (5-HT) TCB-2 TFMFly Tryptamines (e.g., 5-BT , 5-CT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )
Antagonists: 5-I-R91150 5-MeO-NBpBrT AC-90179 Adatanserin Altanserin Antihistamines (e.g., cyproheptadine , hydroxyzine , ketotifen , perlapine )AMDA Atypical antipsychotics (e.g., amperozide , aripiprazole , asenapine , blonanserin , brexpiprazole , carpipramine , clocapramine , clorotepine , clozapine , fluperlapine , gevotroline , iloperidone , lurasidone , melperone , mosapramine , ocaperidone , olanzapine , paliperidone , quetiapine , risperidone , sertindole , zicronapine , ziprasidone , zotepine )Chlorprothixene Cinanserin CSP-2503 Deramciclane Dotarizine Eplivanserin Ergolines (e.g., amesergide , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )Fananserin Flibanserin Glemanserin Irindalone Ketanserin KML-010 Landipirdine LY-393558 mCPP Medifoxamine Metitepine (methiothepin) MIN-117 (WF-516) Naftidrofuryl Nantenine Nelotanserin Opiranserin (VVZ-149) Pelanserin Phenoxybenzamine Pimavanserin Pirenperone Pizotifen Pruvanserin Rauwolscine Ritanserin Roluperidone S-14671 Sarpogrelate Serotonin antagonists and reuptake inhibitors (e.g., etoperidone , hydroxynefazodone , lubazodone , mepiprazole , nefazodone , triazoledione , trazodone )SR-46349B TGBA01AD Teniloxazine Temanogrel Tetracyclic antidepressants (e.g., amoxapine , aptazapine , esmirtazapine , maprotiline , mianserin , mirtazapine )Tricyclic antidepressants (e.g., amitriptyline )Typical antipsychotics (e.g., chlorpromazine , fluphenazine , haloperidol , loxapine , perphenazine , pimozide , pipamperone , prochlorperazine , setoperone , spiperone , spiramide , thioridazine , thiothixene , trifluoperazine )Volinanserin Xylamidine Yohimbine
5-HT2B
Agonists: 4-Methylaminorex Aminorex Amphetamines (e.g., chlorphentermine , cloforex , dexfenfluramine , fenfluramine , levofenfluramine , norfenfluramine )BW-723C86 DOx (e.g., DOB , DOC , DOI , DOM )Ergolines (e.g., cabergoline , dihydroergocryptine , dihydroergotamine , ergotamine , methylergometrine (methylergonovine) , methysergide , pergolide )Lorcaserin MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )Piperazines (e.g., TFMPP )PNU-22394 Ro60-0175 Serotonin (5-HT) Tryptamines (e.g., 5-BT , 5-CT , 5-MT , α-Me-5-HT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )
Antagonists: Agomelatine Atypical antipsychotics (e.g., amisulpride , aripiprazole , asenapine , brexpiprazole , cariprazine , clozapine , N-desalkylquetiapine (norquetiapine) , N-desmethylclozapine (norclozapine) , olanzapine , pipamperone , quetiapine , risperidone , ziprasidone )Cyproheptadine EGIS-7625 Ergolines (e.g., amesergide , bromocriptine , lisuride , LY-53857 , LY-272015 , mesulergine )Ketanserin LY-393558 mCPP Metadoxine Metitepine (methiothepin) Pirenperone Pizotifen Propranolol PRX-08066 Rauwolscine Ritanserin RS-127445 Sarpogrelate SB-200646 SB-204741 SB-206553 SB-215505 SB-221284 SB-228357 SDZ SER-082 Tegaserod Tetracyclic antidepressants (e.g., amoxapine , mianserin , mirtazapine )Trazodone Typical antipsychotics (e.g., chlorpromazine )TIK-301 Yohimbine
5-HT2C
Agonists: 2Cs (e.g., 2C-B , 2C-E , 2C-I , 2C-T-2 , 2C-T-7 , 2C-T-21 )5-Methoxytryptamines (5-MeO-DET , 5-MeO-DiPT , 5-MeO-DMT , 5-MeO-DPT , 5-MT )α-Alkyltryptamines (e.g., 5-Cl-αMT , 5-Fl-αMT , 5-MeO-αET , 5-MeO-αMT , α-Me-5-HT , αET , αMT )A-372159 AL-38022A Alstonine CP-809101 Dimemebfe DOx (e.g., DOB , DOC , DOI , DOM )Ergolines (e.g., ALD-52 , cabergoline , dihydroergotamine , ergine (LSA) , ergotamine , lisuride , LA-SS-Az , LSB , LSD , LSD-Pip , LSH , LSP , pergolide )Flumexadol Lorcaserin MDxx (e.g., MDA (tenamfetamine) , MDMA (midomafetamine) , MDOH , MMDA )MK-212 ORG-12962 ORG-37684 Oxaflozane PHA-57378 Phenethylamines (e.g., lophophine , mescaline )Piperazines (e.g., aripiprazole , BZP , mCPP , quipazine , TFMPP )PNU-22394 PNU-181731 Ro60-0175 Ro60-0213 Serotonin (5-HT) Tryptamines (e.g., 5-BT , 5-CT , bufotenin , DET , DiPT , DMT , DPT , psilocin , psilocybin , tryptamine )Vabicaserin WAY-629 WAY-161503 YM-348
Antagonists: Adatanserin Agomelatine Atypical antipsychotics (e.g., asenapine , clorotepine , clozapine , fluperlapine , iloperidone , melperone , olanzapine , paliperidone , quetiapine , risperidone , sertindole , ziprasidone , zotepine )Captodiame CEPC Cinanserin Cyproheptadine Deramciclane Desmetramadol Dotarizine Eltoprazine Ergolines (e.g., amesergide , bromocriptine , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )Etoperidone Fluoxetine FR-260010 Irindalone Ketanserin Ketotifen Latrepirdine (dimebolin) Medifoxamine Metitepine (methiothepin) Nefazodone Pirenperone Pizotifen Propranolol Ritanserin RS-102221 S-14671 SB-200646 SB-206553 SB-221284 SB-228357 SB-242084 SB-243213 SDZ SER-082 Tedatioxetine Tetracyclic antidepressants (e.g., amoxapine , aptazapine , esmirtazapine , maprotiline , mianserin , mirtazapine )TIK-301 Tramadol Trazodone Tricyclic antidepressants (e.g., amitriptyline , nortriptyline )Typical antipsychotics (e.g., chlorpromazine , loxapine , pimozide , pipamperone , thioridazine )Xylamidine
5-HT3 –7
5-HT3
Agonists: Alcohols (e.g., butanol , ethanol (alcohol) , trichloroethanol )m-CPBG Phenylbiguanide Piperazines (e.g., BZP , mCPP , quipazine )RS-56812 Serotonin (5-HT) SR-57227 SR-57227A Tryptamines (e.g., 2-Me-5-HT , 5-CT , bufotenidine (5-HTQ) )Volatiles/gases (e.g., halothane , isoflurane , toluene , trichloroethane )YM-31636
Antagonists: Alosetron Anpirtoline Arazasetron AS-8112 Atypical antipsychotics (e.g., clozapine , olanzapine , quetiapine )Azasetron Batanopride Bemesetron (MDL-72222) Bupropion Cilansetron CSP-2503 Dazopride Dolasetron Galanolactone Granisetron Hydroxybupropion Lerisetron Memantine Ondansetron Palonosetron Ramosetron Renzapride Ricasetron Tedatioxetine Tetracyclic antidepressants (e.g., amoxapine , mianserin , mirtazapine )Thujone Tropanserin Tropisetron Typical antipsychotics (e.g., loxapine )Volatiles/gases (e.g., nitrous oxide , sevoflurane , xenon )Vortioxetine Zacopride Zatosetron
5-HT4
5-HT5A
5-HT6
Agonists: Ergolines (e.g., dihydroergocryptine , dihydroergotamine , ergotamine , lisuride , LSD , mesulergine , metergoline , methysergide )Hypidone Serotonin (5-HT) Tryptamines (e.g., 2-Me-5-HT , 5-BT , 5-CT , 5-MT , Bufotenin , E-6801 , E-6837 , EMD-386088 , EMDT , LY-586713 , N-Me-5-HT , ST-1936 , tryptamine )WAY-181187 WAY-208466
Antagonists: ABT-354 Atypical antipsychotics (e.g., aripiprazole , asenapine , clorotepine , clozapine , fluperlapine , iloperidone , olanzapine , tiospirone )AVN-101 AVN-211 AVN-322 AVN-397 BGC20-760 BVT-5182 BVT-74316 Cerlapirdine EGIS-12,233 GW-742457 Idalopirdine Ketanserin Landipirdine Latrepirdine (dimebolin) Masupirdine Metitepine (methiothepin) MS-245 PRX-07034 Ritanserin Ro 04-6790 Ro 63-0563 SB-258585 SB-271046 SB-357134 SB-399885 SB-742457 Tetracyclic antidepressants (e.g., amoxapine , mianserin )Tricyclic antidepressants (e.g., amitriptyline , clomipramine , doxepin , nortriptyline )Typical antipsychotics (e.g., chlorpromazine , loxapine )
5-HT7
Antagonists: Atypical antipsychotics (e.g., amisulpride , aripiprazole , asenapine , brexpiprazole , clorotepine , clozapine , fluperlapine , olanzapine , risperidone , sertindole , tiospirone , ziprasidone , zotepine )Butaclamol DR-4485 EGIS-12,233 Ergolines (e.g., 2-Br-LSD (BOL-148) , amesergide , bromocriptine , cabergoline , dihydroergotamine , ergotamine , LY-53857 , LY-215,840 , mesulergine , metergoline , methysergide , sergolexole )JNJ-18038683 Ketanserin LY-215,840 Metitepine (methiothepin) Ritanserin SB-258719 SB-258741 SB-269970 SB-656104 SB-656104A SB-691673 SLV-313 SLV-314 Spiperone SSR-181507 Tetracyclic antidepressants (e.g., amoxapine , maprotiline , mianserin , mirtazapine )Tricyclic antidepressants (e.g., amitriptyline , clomipramine , imipramine )Typical antipsychotics (e.g., acetophenazine , chlorpromazine , chlorprothixene , fluphenazine , loxapine , pimozide )Vortioxetine