|
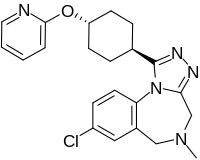
Balovaptan
Balovaptan (INN; developmental code name RG7314), is a selective small molecule antagonist of the vasopressin V1A receptor which is under development by Roche for the treatment of post-traumatic stress disorder.[1] Clinical studiesPost-traumatic stress disorderIt was in a phase III clinical trial for adults and a phase II clinical trial for children for this indication.[2] AutismIn January 2018, Roche announced that the US Food and Drug Administration (FDA) had granted breakthrough therapy designation for balovaptan in people with autism spectrum disorder (ASD).[3] The FDA granted this based on the results of the adult phase II clinical trial called VANILLA (Vasopressin ANtagonist to Improve sociaL communication in Autism) study.[4] The phase III adult study (March 2020) is called V1aduct and the phase II child study is called Av1ation.[citation needed] The phase III study concluded that balovaptan did not improve social communication in autistic adults.[5] It was also in phase II studies for the treatment of stroke. However, it has since been discontinued for both of those indications, and is only being developed for post-traumatic stress disorder, with plans to submit regulatory filings in 2025 or later.[6] References
|
||||||||||||||||||||||||||||||||||||||||